How the Brain Mounts Its Defence in Acute Injury Comes into View
A collaboration between the research groups of Ádám Dénes (HUN-REN Institute of Experimental Medicine) and Jing Miao (CAS Brain Institute, Shanghai) has revealed how the brain regulates the strength and extent of its responses to acute injury. Using next-generation biosensors and real-time imaging methods, the researchers were able to monitor the molecular processes through which ATP can play a central role as an injury signal in directing neuroimmune mechanisms. The findings may help us understand how the brain recognises endangered neurons and how it organises a local defensive response in the event of acute damage.
Ádám Dénes’s research group discovered that microglia form functional connections with neuronal cell bodies through ATP sensing, and that in brain infections they also identify affected neurons on the basis of local ATP release. In the brain, ATP therefore serves not only as a source of cellular energy, but also as one of the most important injury-signalling molecules. The paper, now published in Neuron, one of the most prestigious international journals in neuroscience, examines how the brain controls the magnitude of the response to acute injury in space and time. ATP may again be a central player in these processes.
To investigate the injury-sensing capacity of microglia, Ádám Dénes and Jing Miao were awarded a Chinese-Hungarian bilateral research grant in 2020.
The G protein-coupled receptor (GPCR)-based next-generation biosensors developed in Jing Miao’s laboratory and further refined through the collaboration were optimised for real-time imaging measurements of ATP in the brain. These GPCR-based sensors make it possible to visualise the release of different purinergic mediators in the brain parenchyma and in the perivascular environment with previously unimaginable nanomolar sensitivity and are therefore suitable for investigating neuroimmune interactions and pathological processes in the nervous system.
A 2024 study by Miao’s research group, published in Nature Neuroscience, showed that in the event of focal injury, astrocytes release rhythmic ATP clouds, which are capable of recruiting microglial processes even at concentrations amounting to only one hundred-thousandth of the intracellular ATP concentration. It was not clear, however, how the microglial response feeds back on ATP release by astrocytes, or what effect this has on the development of brain injury or neuronal network disturbances following insult. The researchers found that the pro-inflammatory cytokine interleukin-1 beta (IL-1β), released by microglial cells, regulates astrocytic ATP release through negative feedback, thereby limiting the spatiotemporal extent of brain injury. This is particularly noteworthy because global inhibition of IL-1β improves outcome in most models of brain injury, including traumatic brain injury and stroke, according to clinical data as well.
It also became clear that the severity of the injury is a determining factor: in the case of brain microinjuries, microglial IL-1β production is likely to be an evolutionarily conserved and beneficial process that restricts the spread of injury, whereas in larger brain injuries or during inflammation this balance may be disrupted, inhibiting or even reversing the effectiveness of the negative feedback. The article describes this broader mechanism as a reciprocal microglia-astrocyte feedback circuit that helps calibrate injury information and maintain the tissue response balance.
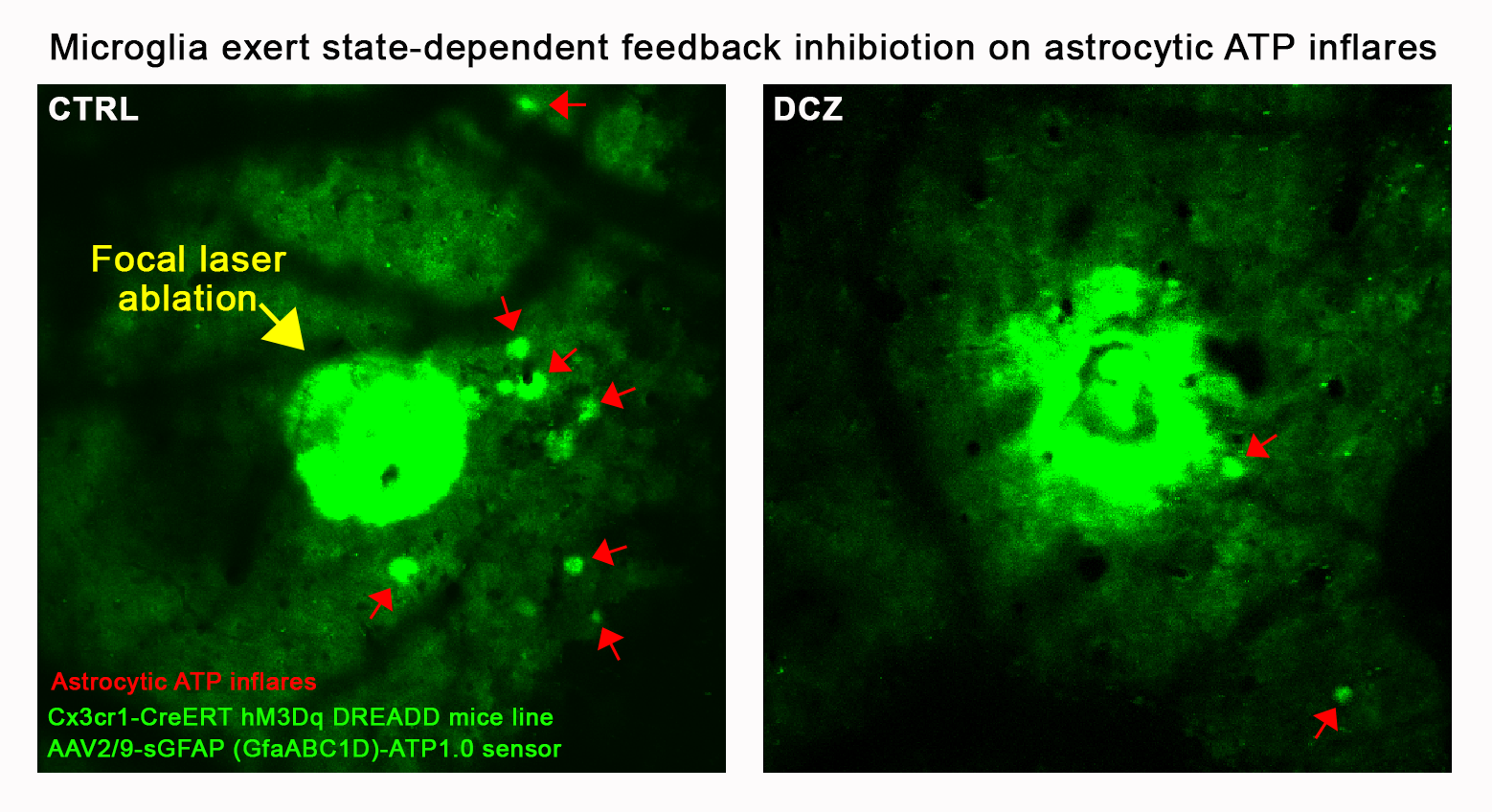
The figure shows an experiment performed in a chemogenetically modified mouse model, in which microglial cells can be selectively activated by DCZ administration, while astrocytic ATP release events are monitored using virus-mediated labeling. Following focal laser ablation, extensive astrocytic ATP release is observed in the broader vicinity of the lesion under resting microglial conditions. In contrast, microglial activation markedly reduces the number of these ATP release events. These results suggest that microglia regulate astrocytic ATP release through a state-dependent negative feedback mechanism.
On the basis of the jointly developed concept, most of the experimental work was carried out in the Chinese laboratory, while key microglial manipulation and validation experiments were performed in the Dénes laboratory. Zsuzsa Környei showed in several models that both the mere presence of microglia and the addition of IL-1β limit the development of astrocyte-associated ATP events, while Anett Schwarcz, using in vivo two-photon microscopy, confirmed the ATP-dependent dynamics of microglia-astrocyte interactions with the aid of the MicroDREADD mouse line developed in the laboratory.
Without real-time imaging, it is not possible to understand the complexity of brain injury and immune processes, and in this respect—particularly in the case of simple, rapidly metabolised molecules such as ATP—the new generation of GPCR-based biosensors has already enabled major scientific discoveries. Their application represents a further exciting and potentially surprising avenue in the investigation of neuroimmune processes in the brain.