Hungarian Researchers Offer New Explanation for One of Evolution’s Greatest Mysteries: Why Eukaryotic Cells Evolved Only Once
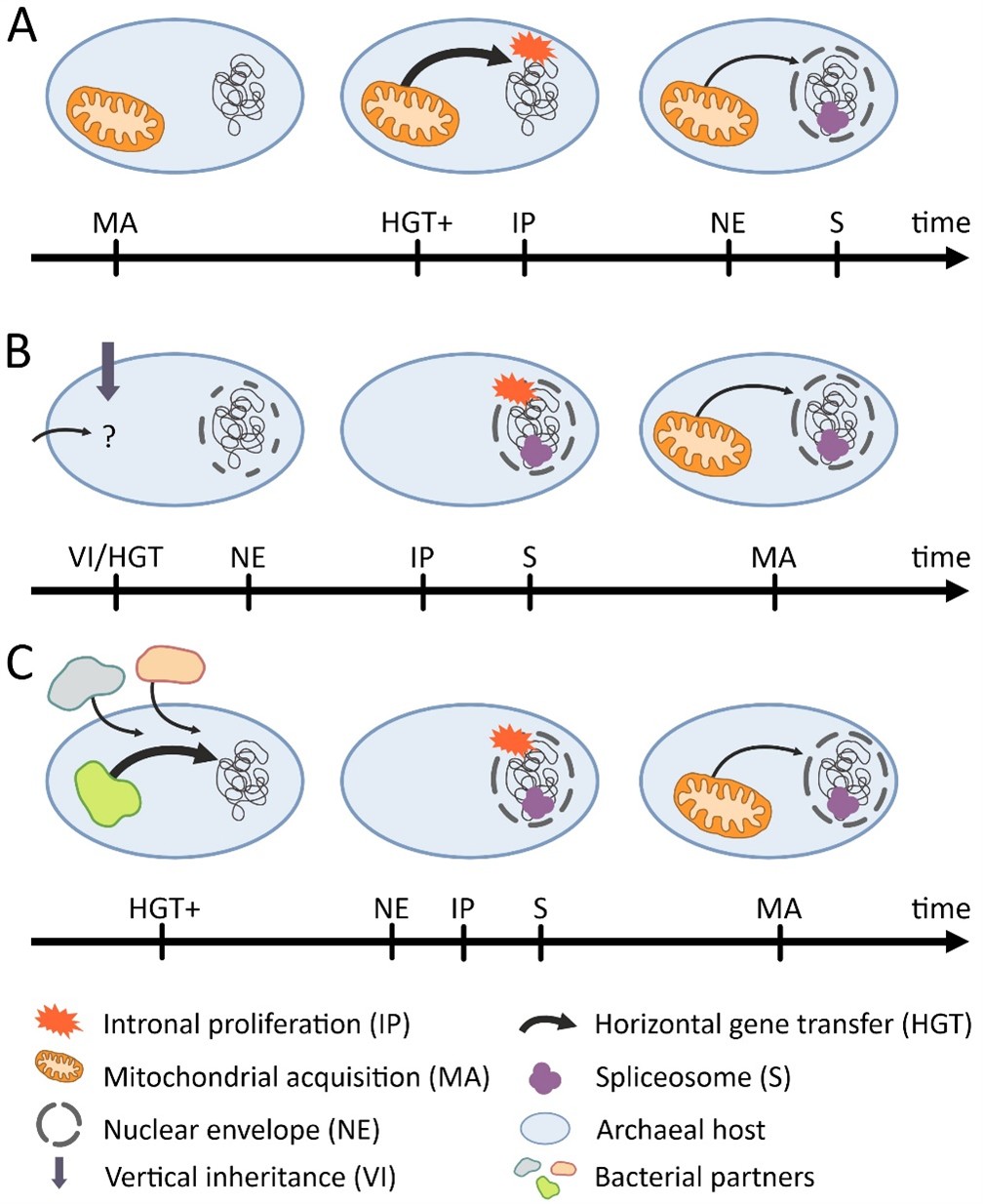
In their study published in a special issue of Philosophical Transactions of the Royal Society, István Zachar, Jakab Máté and István Oszoli, researchers at the HUN-REN Institute of Evolution, offer a new perspective on the origin of the nucleus and the emergence of eukaryotes. Although both the nucleus and the mitochondrion are unique and complex eukaryotic innovations, conventional explanations suggest that the long process of eukaryogenesis began when the bacterial partner regarded as the ancestor of the mitochondrion was engulfed by a host cell. The Hungarian researchers argue instead that the crucial step may have been the emergence of the nucleus itself, which may have started to evolve before the mitochondrion and may have paved the way for the first stable intracellular endosymbiotic partnership.
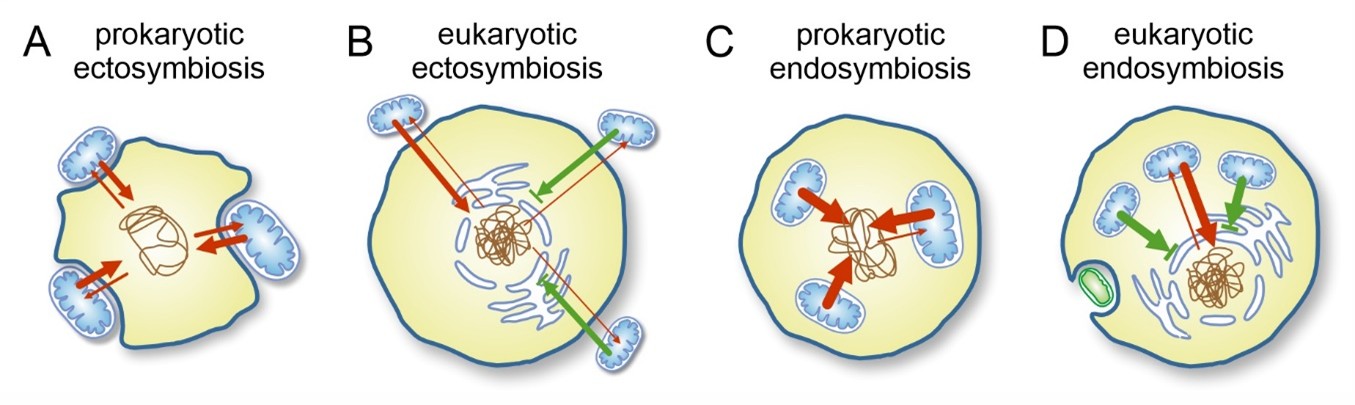
The amount of expected horizontal gene transfer (HGT) under various conditions of symbiosis. Red arrows indicate potential gene transfer, green arrows blocked transfer attempts; the strength of arrows indicates presumed magnitude of successful transfers in unit time.
Horizontal gene transfer (HGT), in which microbes exchange genetic material with one another, is extremely common among unicellular organisms. This process is particularly widespread among prokaryotes, especially among bacteria, and although it allows rapid adaptation, it also creates instability: bacterial genomes constantly change as cells adapt to shifting resources, environments and interaction partners. As a result, close and species-specific cooperative relationships are difficult to maintain over the long term.
Coexistence and cooperation are also widespread among bacteria, yet stable intracellular symbioses seem to be missing. The high frequency of HGT may help explain why stable endosymbiotic partnerships are absent in prokaryotes: there is simply too much genetic permeability, and the host’s genetic material is under constant threat as alien DNA continuously interferes with it. Moreover, symbiotic partners (even if they do enter the cell) can easily be replaced or exploited: once useful genes have been acquired, the host may dispense with the partner.
According to the authors, the nucleus or some less evolved endomembrane system that may have preceded it could have offered a solution to this problem. By sequestering DNA behind extra membranes and spatially separating transcription from translation, the host could have reduced the likelihood that foreign (potentially harmful) DNA is transcribed and incorporated. Once the nuclear envelope had evolved, the next engulfed bacterium (the ancestor of all modern mitochondria) would have been far less harmful to the host genome. Internal compartmentalization of DNA enabled safer transfer and integration of beneficial symbiont genes into the host genome at a slower pace. In this way, gene flow became increasingly controlled by the host, making stable endosymbiosis possible. In the authors’ view, the key to the origin of eukaryotes was therefore not endosymbiosis itself, but the preceding internal structuring and spatial division of labour. This unique innovation may explain why this evolutionary breakthrough happened only once.