The Macrophage Polarization Group established three months ago at the Institute of Genetics of the ELKH Biological Research Centre (BRC) with the leadership of Zsolt Czimmerer), continuing Zsolt’s former research at the University of Debrecen, are investigating the transcriptional and epigenetic background of macrophage polarization and the memory-like properties of innate immune cells in collaboration with researchers from several national and international laboratories. Zsolt Czimmerer’s latest related study was published in the prestigious journal Immunity.

Macrophages are highly plastic and heterogeneous cellular elements of the innate immune system. Their function and phenotype are significantly influenced by microenvironmental signals that trigger various epigenetic and gene expression programs through the activation of transcription factors, thereby affecting the induced macrophage polarization pathway. The research group formed in September 2022 aims to investigate the effects of the microenvironment on macrophage function and phenotype by combining methods from epigenetics, immunology and transcriptomic technology. Researchers are also looking at how the simultaneous or temporally divergent presence of several different microenvironmental signals regulates gene expression within macrophages and hence polarization.
In the paper presenting the results the researchers identified new synergistic interactions between alternative macrophage polarization and signals of different pathogenic origin, in addition to the previously known interactions that mutually inhibit one another, and which confer novel functional properties to macrophages. Using complex epigenomic, transcriptomic and immunological approaches they found that the intense epigenetic reprogramming during alternative macrophage polarization results in a significant increase in complex binding of transcription factors activated by pathogen-derived molecules at the whole genome level. Increased transcription factor binding is associated with increased activity of genomic regulatory elements, leading to synergistic gene activation. Finally, the researchers also confirmed the existence of the identified phenomenon in a mouse model of allergic asthma.
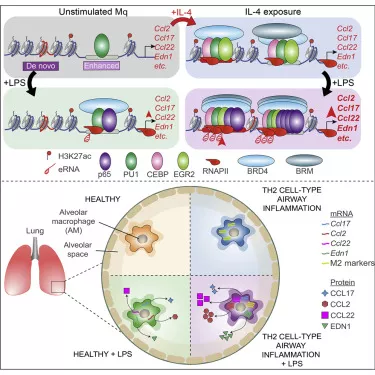
This discovery can help to understand the pathomechanisms of complex diseases in which opposing polarization signals are simultaneously present in the macrophage microenvironment, co-regulating their function and significantly influencing disease outcome. The results of this research could also play an important role in identifying new therapeutic approaches in the future for severe and difficult-to-treat conditions such as glucocorticoid-resistant ‘mixed’ asthma.
The Macrophage Polarization Group works together with colleagues from several national and international laboratories. With László Nagy (University of Debrecen, Johns Hopkins University), Attila Bácsi (University of Debrecen), Bence Dániel (Stanford University), and Marten Hoeksema (University of Amsterdam) they are investigating the transcriptional and epigenetic background of macrophage polarization. With Jolán Walter and Krisztián Csomós (University of South Florida) they are collaborating to map the epigenetic and transcriptional regulatory mechanisms involved in the generation of ABC (age-associated B cells). Joint projects with Gábor Szebeni and Viktor Honti from BRC have begun on memory properties of innate immune cells.